Transfusion-transmissible infection surveillance report
Transfusion-transmissible infections surveillance reports
The 2025 transfusion-transmissible infections in Australia surveillance report has now been published and the major findings are as follows:
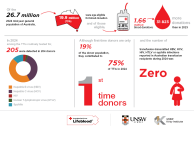
1. A total of 205 transfusion-transmissible infections (TTIs) were detected in 204 blood donors for which testing is in place: hepatitis B virus (HBV), hepatitis C virus (HCV), human immunodeficiency virus (HIV), human T-lymphotropic virus (HTLV), or potentially infectious syphilis. One donor was positive for both hepatitis C and HIV.
2. Consistent with the long‑term pattern, the most common TTI detected was hepatitis B, followed by hepatitis C.
3. Although representing 19% of the donor population, first-time blood donors in 2024 contributed to 75% of detected TTIs, lower than the 85% observed in 2023.
4. Supporting the effectiveness of donor education and selection, the prevalence of TTIs in 2024 is substantially lower among first-time blood donors (12 to 187 times) than in the general population in 2023/2024.
5. The estimated residual risk of transmission for hepatitis B, hepatitis C, HIV, HTLV and syphilis in Australia is very low – less than one in one million per unit transfused. This supports that Australia’s blood supply is among the safest worldwide in respect of TTIs for which testing is conducted. Despite this, there remains a minimal but real risk of TTIs that must be carefully considered before any transfusion.
6. The non-compliance rate among TTI-positive donors in 2024 was 27%, at the upper range of the 15-27% observed in the last decade. The current rate highlights the importance of promoting donor education to ensure that potential donors understand the importance of ‘self-deferral’ to reduce the risk of collecting blood from a potentially infected donor whose infection may not be detected by testing.
7. There were four confirmed cases of transfusion‑transmitted bacterial infection. Two each, with Bacillus hominis (from one platelet donation) and Staphylococcus aureus identified. The number of TTBIs and near-miss events during 2024 was highly unusual and was subject to an extensive investigation with regular reporting to the Therapeutic Goods Administration. There were no quality or procedural deviations. Several potentially causal factors were considered with no definitive cause identified. No further cases have been reported in 2025 up until the date of publication of this report.
8. There were two hepatitis E transfusion‑transmissions (from a single donor). In both cases, infection resolved without ongoing complications.
9. No probable or confirmed transfusion‑transmitted hepatitis B, hepatitis C, HIV, HTLV or syphilis cases were reported in Australia during 2024.
Updated December 2025
For older versions of the Transfusion-transmissible infections surveillance reports and infographic
please view the Resource Library.